Entrepreneurial Support Program(Explore Course)

- Principal Investigator
-
University of Tsukuba
Hidetoshi Takada
- Adopted Theme
Development of an Innovative mRNA Vaccine Platform Technology to Control Autoimmune Diseases
- Subject of Research
- Development of an Innovative mRNA Vaccine Platform Technology to Control Autoimmune Diseases
- GTIE VC Collective
Mitsubishi UFJ Capital Co., Ltd.
Angel Bridge Inc.
- Overview
-
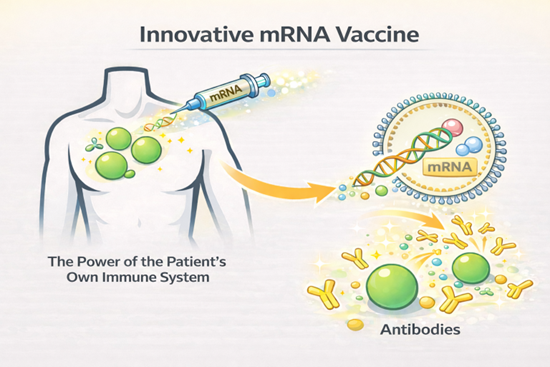
Currently, the treatment of autoimmune diseases is predominantly based on high-cost antibody therapeutics. In Japan, annual treatment expenses can reach several million yen per patient, while in the United States they may amount to tens of millions of yen per year. This represents a substantial burden on both patients and healthcare systems. As the sustainability of healthcare expenditures has become a global challenge, there is a strong need to establish new therapeutic platforms from a long-term perspective.
In this project, we aim to develop a next-generation therapeutic approach that induces sustained antibody production by leveraging our proprietary mRNA vaccine platform technology and harnessing the patient’s own immune mechanisms. As the initial target, we have selected a specific area within autoimmune diseases. In this field, the treatment market, centered on biologics, continues to expand and is projected to reach approximately JPY 10 trillion (around USD 70 billion) by 2032, with a compound annual growth rate (CAGR) exceeding 10%. Our technology seeks to provide a novel therapeutic option that achieves both high efficacy and cost-effectiveness within this growing market.
Furthermore, this technology is expected to evolve beyond a single indication into a versatile drug discovery platform applicable to multiple autoimmune diseases. By enabling both healthcare cost optimization and improvements in patients’ quality of life (QOL), this project is positioned as a research and development initiative of significant societal value.
Key Features of the Technology
・By harnessing the patient’s own immune mechanisms, the technology induces an antibody response that is expected to maintain stable and high efficacy over an extended period.
・It aims to establish a therapeutic design that reduces dosing frequency, thereby improving patients’ quality of life (QOL) while also contributing to the optimization of healthcare costs.
・Beyond a single disease, the technology is expected to serve as a drug discovery platform applicable to multiple autoimmune diseases.
- Business Models(when applying)
This business does not assume in-house commercialization as a prerequisite. As its basic strategy, the program will be out-licensed to global pharmaceutical companies after obtaining human proof of concept (POC) in early-stage clinical trials (Phase I/II).
Revenue will be secured through a drug discovery–based business model, with milestone payments corresponding to each stage of development and royalties following market launch.
- Activity Planning(when applying)
・Therapeutic efficacy will be evaluated in disease models using humanized model mice, and development candidates will be selected accordingly.
・Efficacy and safety data will be obtained through studies in humanized mice and non-human primates, and proof of concept (POC) will be established at the non-clinical stage.
・In parallel, market and competitive analyses will be conducted, and a business plan capable of supporting investment decisions will be formulated.
・Upon completion of the program, a startup company will be established with the aim of initiating Phase I/IIa clinical trials.
・Through dialogue with global pharmaceutical companies and data-driven analyses, the market size and competitive landscape will be organized, and mid- to long-term revenue models and capital strategies will be developed.
・An integrated management structure combining research, business, and finance will be established to steadily promote social implementation.
