Entrepreneurial Support Program(Oversea market pioneer practice Course)

- Principal Investigator
-
The University of Tokyo
Makoto Kondo
- Adopted Theme
Restoring Microvascular Health by Enhancing Vascular Resilience
- Subject of Research
- Restoring Microvascular Health by Enhancing Vascular Resilience
- GTIE VC Collective
UTokyo Innovation Platform Co., Ltd. (UTokyo IPC)
- Overview
Diseases caused by vascular dysfunction remain areas of significant unmet medical need, with limited effective treatment options available in clinical practice. This project aims to create a startup that delivers a novel vascular regenerative factor modality to medical institutions, leveraging three-dimensional culture systems and advanced analytical technologies. By establishing non-clinical proof of concept while simultaneously advancing development with regulatory and CMC considerations in mind, we seek to enable highly translational therapeutic technologies ready for clinical application. Furthermore, through collaboration with international key opinion leaders (KOLs) and cell banks, this project will lay the foundation for future global expansion and introduce new therapeutic options for vascular-related diseases.
- Business Models(when applying)
We aim to establish a licensing and product supply model through a research-driven startup centered on a proprietary vascular regenerative factor modality. Initially targeting refractory ischemic diseases, revenue generation will be pursued through co-development and licensing agreements with pharmaceutical companies and related partners. To establish a de facto global standard in vascular regenerative medicine, we will strategically secure intellectual property and advance a product development framework aligned with international clinical needs.
- Activity Planning(when applying)
-
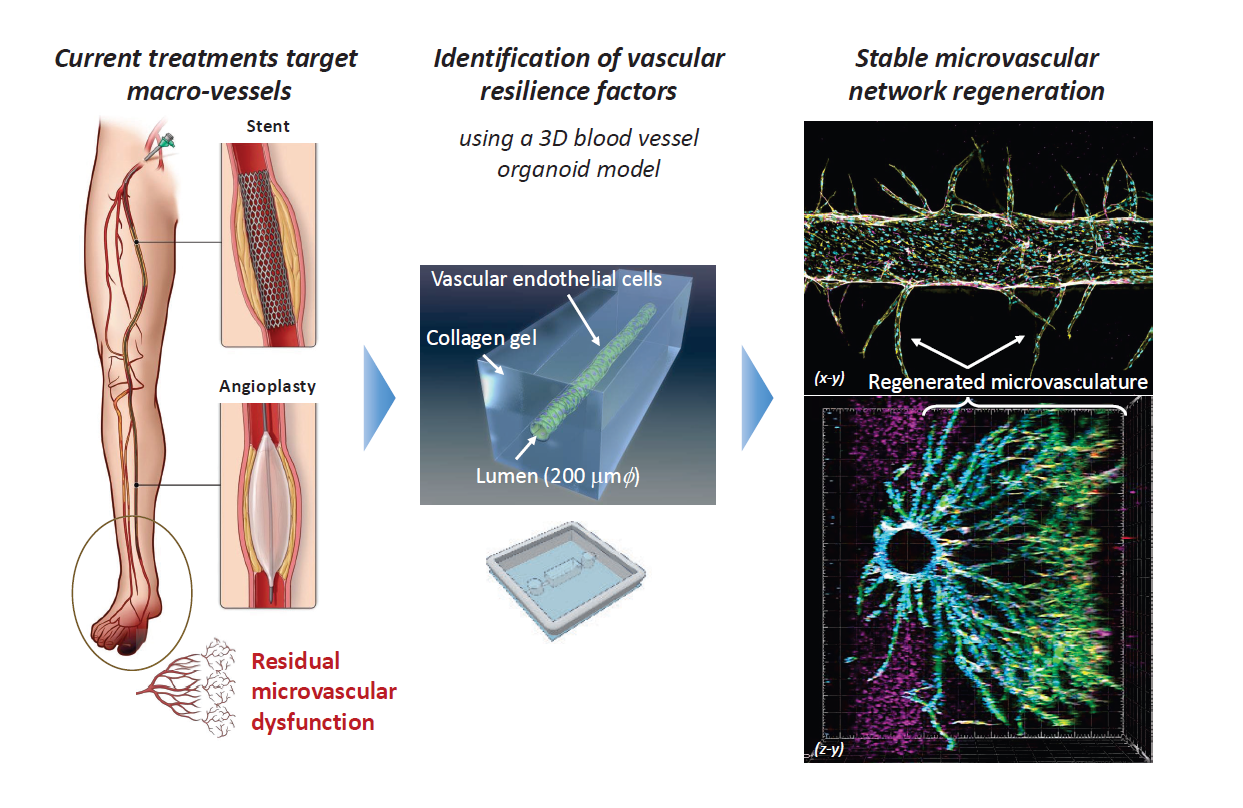
We will prioritize the generation of candidate regenerative factors using three-dimensional culture systems and the acquisition of non-clinical PoC through in vitro and in vivo studies. In parallel, regulatory strategies (Pre IND/Q sub) and CMC design will be examined to clarify feasible and scalable product configurations for clinical application. Furthermore, through collaboration with international KOLs and cell banks, we will develop an integrated R&D and commercialization framework designed to support global expansion.
