Entrepreneurial Support Program(Explore Course)

- Principal Investigator
-
Yokohama National University
Junji Fukuda
- Adopted Theme
Treatment of Male Pattern Hair Loss Using microRNAs Generated by Human Dermal Papilla Cells in a Multi-Layer Culture System
- Subject of Research
- Treatment of Male Pattern Hair Loss Using microRNAs Generated by Human Dermal Papilla Cells in a Multi-Layer Culture System
- GTIE VC Collective
KSP, Inc.
- Overview
-
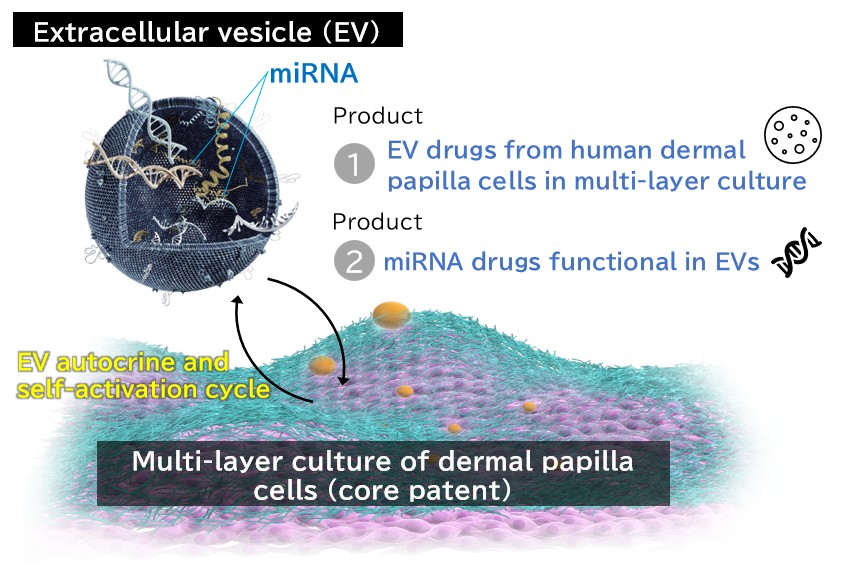
Various drugs and approaches have been investigated for the treatment of androgenetic alopecia, which accounts for the majority of hair loss. While some are used in clinics, their effect is insufficient, leading to a strong demand for new effective treatments. This project proposes extracellular vesicles generated from human dermal papilla cells in the multi-layered culture technology, which is established by the principal investigators. In addition, considering that extracellular vesicles still present challenges in manufacturing, purification, quality standards, and cost, the project will prioritize advancing commercialization of microRNA specifically contained in the extracellular vesicles from human dermal papilla cells.
- Business Models(when applying)
The treatment involves formulating extracellular vesicles produced by dermal papilla cells or the microRNA contained within them, supplying them to medical institutions where they are administered into the scalps of alopecia patients. Treatment will commence domestically, with overseas expansion proceeding while complying with each country's legal regulations. Regarding the exit strategy, once favorable results are confirmed in exploratory clinical trials, negotiations for licensing out to major pharmaceutical companies or M&A will commence.
- Activity Planning(when applying)
The commercialization promotion organization will take the lead in analyzing market research and technology trend survey results to refine the business strategy and establish competitive advantage in the market. Furthermore, using the customer targets and development pipeline set in the initial provisional business plan as hypotheses, it will investigate and analyze detailed market needs to verify whether they can become appropriate solutions in the alopecia field. Concurrently, technology trend surveys and regulatory environment surveys will also be conducted. Research activities will involve synthesizing the identified microRNA, encapsulating it in a lipid capsule for drug delivery, and determining the transdermal administration method. Specifically, the transdermal injection method will be selected from the devices of partner companies possessing medical device-approved injectors. Subsequently, an approval application will be submitted as a nucleic acid-based pharmaceutical, including safety testing.
